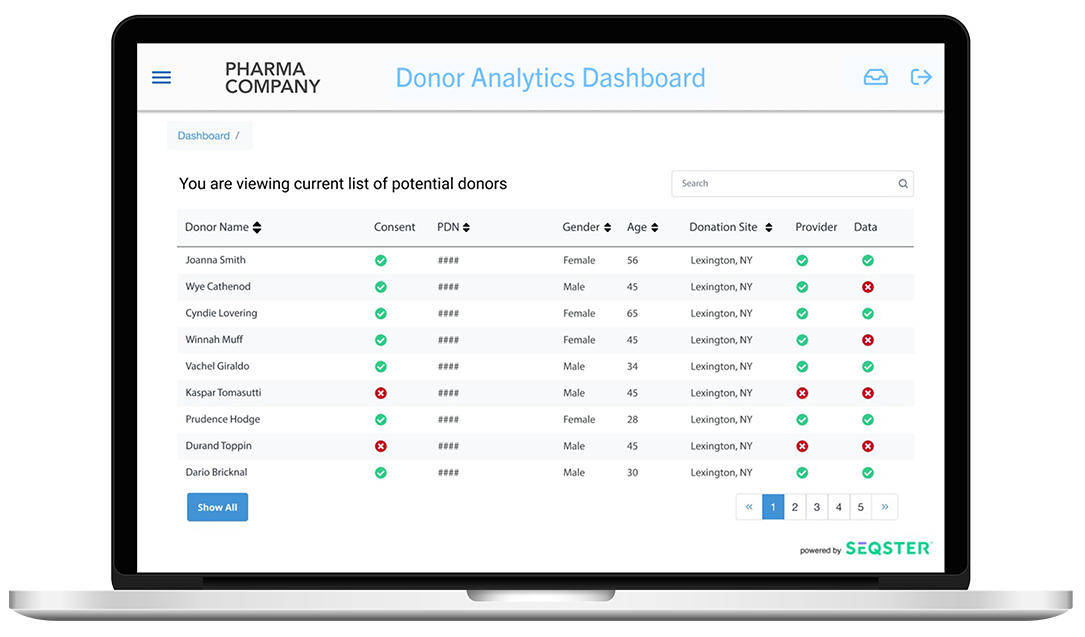
Improve clinical trial success by delivering high quality health data that facilitates patient recruitment and retention.
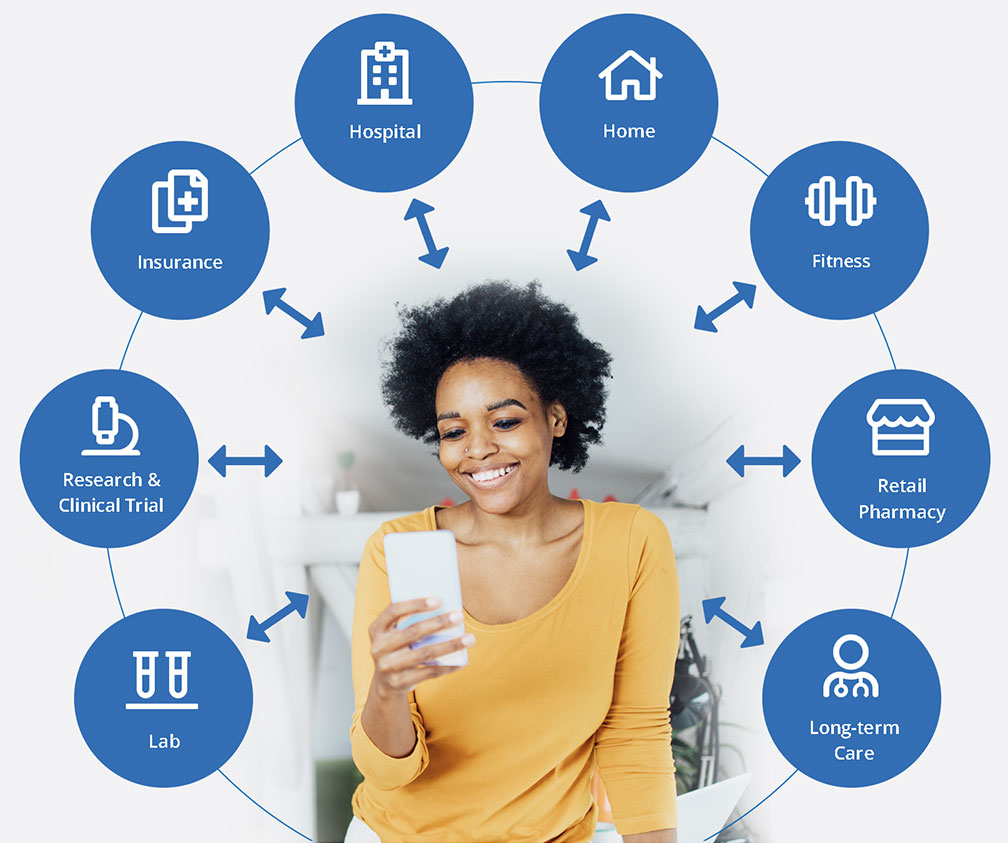
- One unified view of all health, genetic and fitness data organized in a longitudinal record
- The best nationwide health system coverage with pre-built access to 150,000 hospitals and medical groups
- Patient protocol matching with structured data search leveraging all coded terms including SNOMED, ICD10, RxNorm, NCD, CVX and LOINC codes

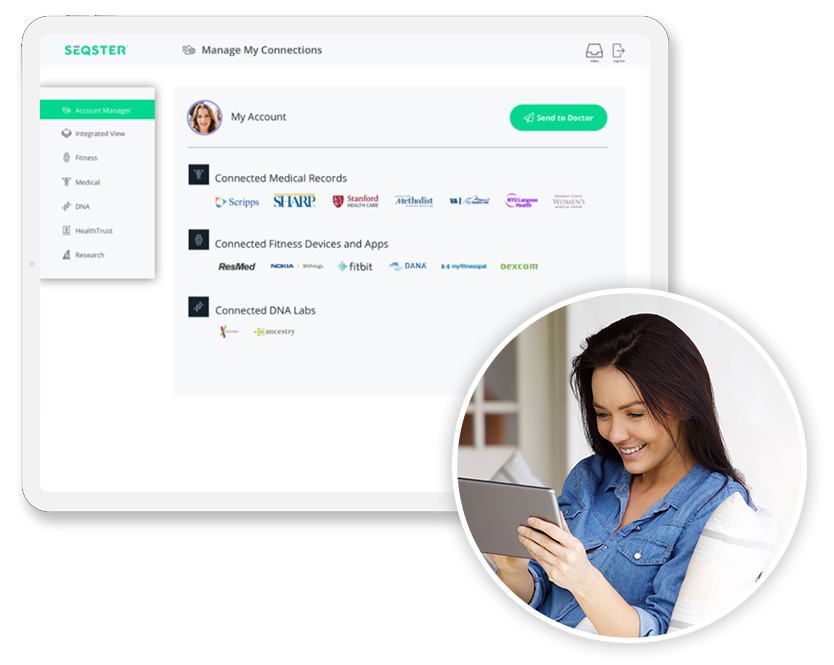
Accelerate clinical trials by automating health data collection and cleansing.
- Rapid go-live with minimal IT involvement using a pre-built, configurable patient portal
- Real-time connection to a patient’s entire medical history through a patient-mediated data aggregator
- Hassle-free web and mobile integration using a UI-driven software development kit (SDK)
Demonstrate a drug’s value by accessing the most comprehensive and real-time set of RWD types for analysis.
- Medical history, genetic risk factors, lifestyle, quality of life and social determinants of health data sources
- Treatment response tracking with continuously updated medical and lab information
- Better decision making by linking patient RWD with any other data sources using the most widely adopted tokenization technology
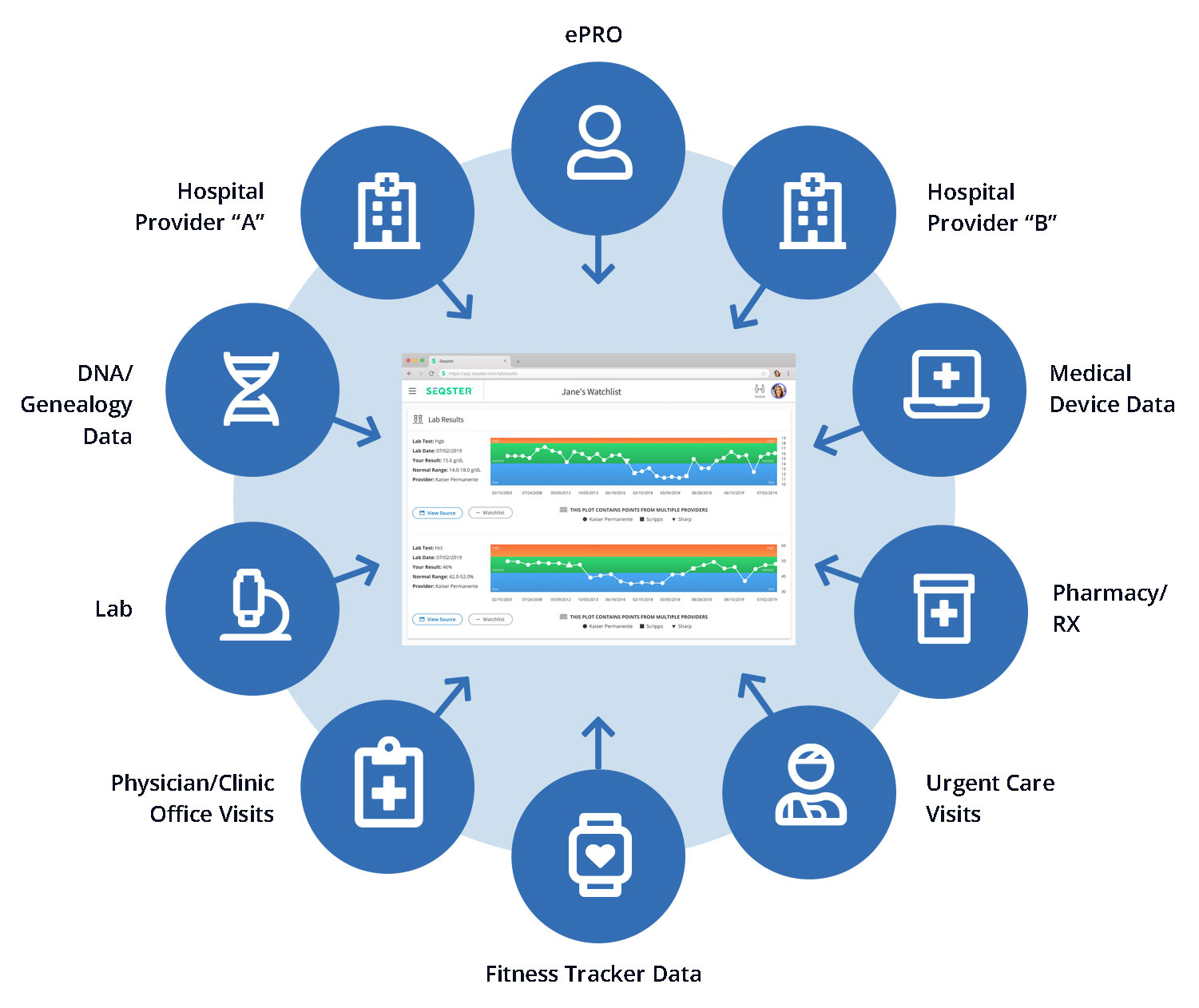

SEQSTER Digital Front Door
The Participant Digital Front Door™ solves a multitude of data challenges for patients enrolling in and researchers conducting clinical trials.
The Digital Front Door places the patient at the center of the trial and their health data to advance science through their contribution to research.
- Health Data Access & Longitudinal Health Profile
- Data Sharing with Caregiver/Family & Physician/Care Team
- Simplifies the Informed Consent Process
- Boosts Participant Engagement leading to Retention
- Alleviates Caregiver Burden


SEQSTER Digital Front Door
For Researchers conducting Clinical Studies & Trials, the Participant Digital Front Door™ powers RWE, HEOR, and Accelerated Drug Discovery through:
- Automated Real-Time Health Data Collection:
Retrospective + Prospective Data - Study Design Flexibility & Control: Site-based + Decentralized + Hybrid
- High Quality & High Fidelity Real World Data: EHR + DNA + Remote Patient Monitoring
- Regulatory Compliance: HIPAA, Interoperability and Patient Access Final Rule, 21st Century Cures Act, and FDA 21 CFR Part 11
- Achieving Greater Health Equity & Trial Diversity
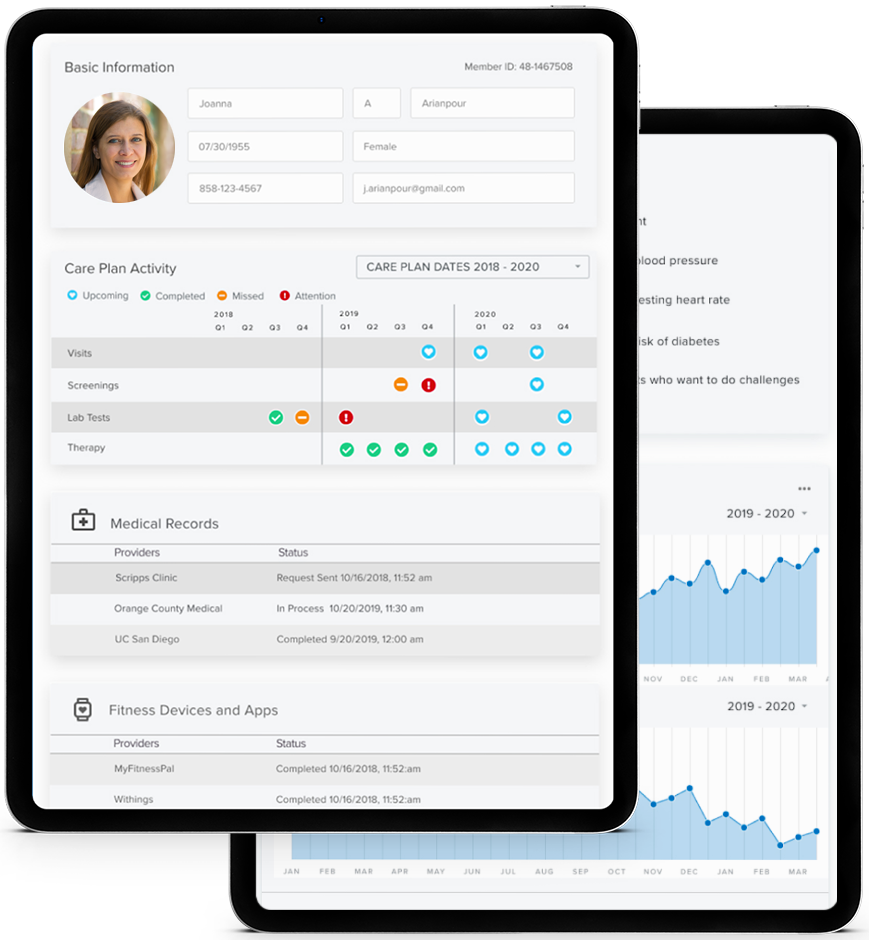
Digital Front Door for Care Management
- Personalize member experience/ navigation
- Facilitate care coordination
- Leverage clinical data analytics to:
- Enable proactive outreach
- Identify care gaps
- Augment patient care planning
- Support value-based contracts
- Improve Risk Adjustment, HEDIS and care management scores
Comprehensive Data + Efficiently Captured = Better Outcomes

We are breaking down data silos to:
- Multidimensional data sources: Genomic/DNA,
Medical Records/EHR, Fitness/Medical Device,
Device, Rx/Pharmacy provide the most comprehensive
source or patient data available - Multimodal EHR retrieval using a
wide range of APIs and aggregators - Commonwell Health Alliance member
API for B2B Partners
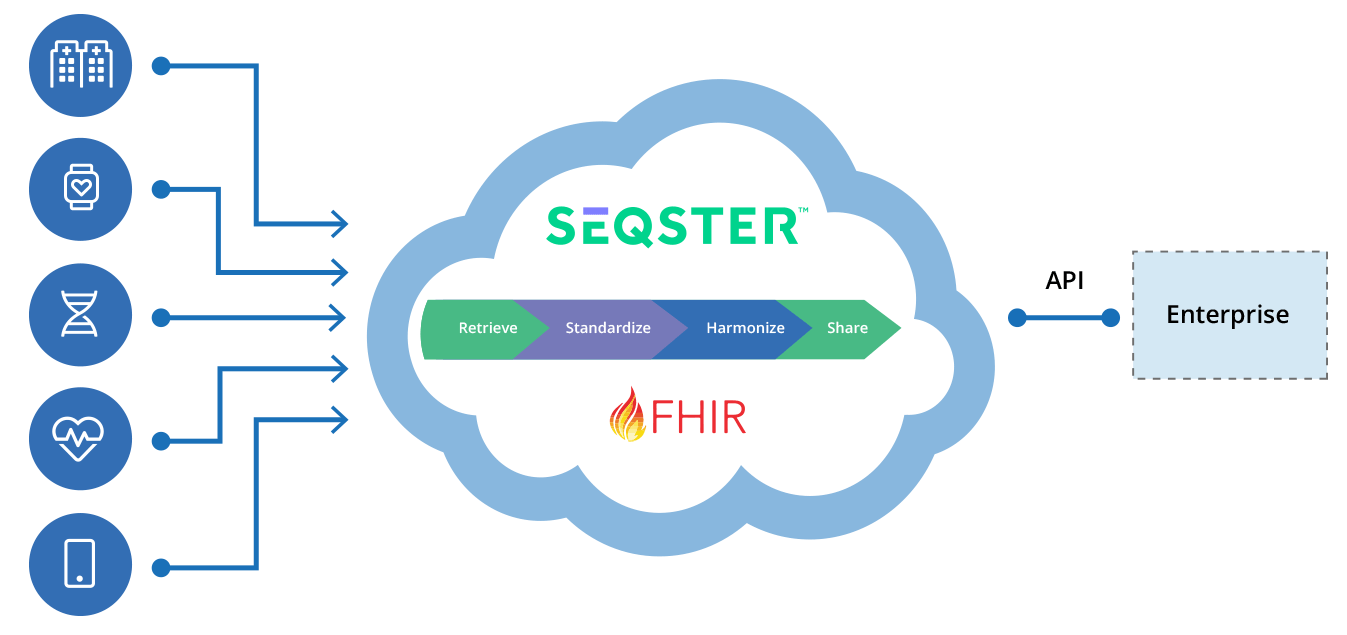
Granular Data Access
- Providers
- Allergies
- Conditions
- Medication
- Doctor Visits
- Immunizations
- Lab Results
- Procedures
- Vitals
- Demographics
Full Data Dump
- Timeline View
- Categorical View
- EHR data is standardized, normalized, and delivered to the Pharma data lake for analytics and machine learning
- By matching clinical data related to the study criteria, SEQSTER can assist in determining eligibility and highlighting terms related to the research
- Continue monitoring patient care post-trial for outcomes analysis
Recruit. Engage. Retain.
SEQSTER drives greater value for clinical trials by delivering patient-consented healthcare data from multiple sources in real-time.
Explore historical and current data to derive insights from symptoms to diagnosis to treatment. Continue retrieving and observing patient data for long-term outcomes tracking.
Are you ready for the SEQSTER experience?